Causes of MS
You may wonder whether you did something to cause your MS or if you could have stopped MS from happening to you. The answer is 'no'. No one could have advised you how to prevent your MS.
The immune system is the body's main defence system against infections or other foreign substances. It involves many different types of cell, organised in complex chains of action. The immune system detects and destroys bacteria, viruses and parasites that enter the body.
At the heart of the system is your body's ability to recognise which cells are part of you, and which are not. Your body cells are all marked with protein flags made by the highly variable human leukocyte antigen (HLA) collection of genes. The cells of your immune system ought to recognise and leave them alone. In autoimmune diseases such as MS, rheumatoid arthritis or lupus, something happens to make the immune system target your own body cells for destruction. In MS, the target is the myelin sheath around the nerves of the central nervous system.
The immune system is highly complex and is a key area for research into new MS treatments. Here is a simple overview of the immune system.
Your body's first line of defence is a simple inflammatory response. Everyone has this defence system from birth, and it is known as the innate immune system.
A pathogen might enter the body through a wound, or through the nose or mouth. It is detected, and damaged cells nearby release histamine. This causes a local swelling as it opens up blood vessels to leak fluid into the area. The swelling makes it easier for other white blood cells to get to the site.
The first immune cells on the scene are often macrophages. These large cells are a kind of phagocyte, and can engulf pathogens and destroy them. This process is called phagocytosis, and is shown below.
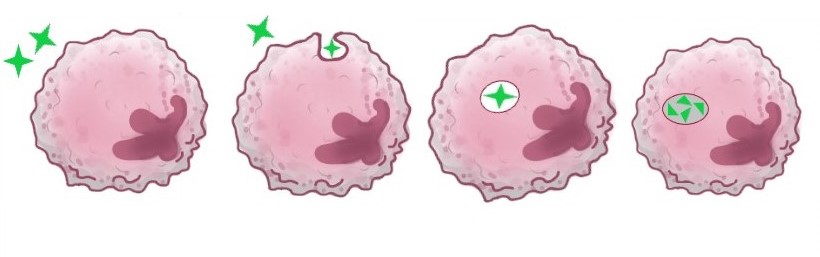
Other kinds of phacogcyte release chemicals as part of the first defence response. Some of these act as signals to encourage further inflammation. Some of these chemicals are toxic and so destroy any of your own cells nearby, as well as the invaders. Other phagocytes can extend long chains of molecules around them which act as a physical net to prevent pathogens moving further through the body. Phacocytes are the major component of the pus formed in a wound.
In a small infection, this response would be sufficient to protect you. If it doesn't resolve the situation quickly, then other cells get involved in a more sophisticated acquired immune response.
Your body can learn how to recognise previous infections, and so be ready to fight them off faster if you are exposed to them again. This is why children are routinely vaccinated against diseases they might encounter later in life. This secondary defence system involves different leukocytes and is known as the acquired immune system. The leukocytes used in the acquired immune system are called lymphocytes, and they include various kinds of T-cell and B-cell.
If an infection is not dealt with quickly by the first line of defence, your lymphocytes get involved. Some of the cells that have engulfed pathogens present fragments of the pathogen on their own cell surface. Lymphocytes called helper T-cells detect the antigens and encourage more inflammation and thus more fluid and blood cells to come to that location. The helper T-cells also activate other lymphocytes called B-cells and killer T-cells, according to the type of threat the body faces.
The B-cells start producing antibodies matched to the antigen. These attach to the antigen to disable it. Killer T-cells are then more likely to find cells that have been infected with the pathogen and destroy them.
Once the crisis is over, helper T-cells start to signal with a chemical called beta interferon, which calms down your immune response.
Most of the cells involved with an immune reaction will die after the infection is over. However, some T-cells and B-cells remain after the infection is cleared up. They can survive for years in the lymph glands or marrow, and act as memory cells. If the same disease is encountered again, your immune response will be swifter and more effective.
The central nervous system (CNS) is generally sealed off from the rest of the body by a membrane layer which prevents large molecules, immune cells or pathogens from crossing into the space around the brain and spinal cord. This is known as the blood-brain barrier (BBB). If you have MS, then immune cells have crossed into the CNS, and somehow become triggered to treat your own myelin as a target antigen.
Each link in the immune response is a point where an MS treatment could potentially intervene. However, because there are many types of immune cell, all with different operations, it is not that easy. Boosting or interrupting one cell function could be counter-acted elsewhere in the chain. The currently licensed disease modifying drugs (DMD) have a variety of different actions, so if you cannot tolerate one, there may be other options.
Several current DMD options are forms of beta interferon (Avonex, Betaferon, Extavia, Plegridy and Rebif). This is the chemical that T-cells release to signal that an immune attack is dealt with and that inflammation can calm down. The intention is that taking extra beta interferon will reduce inflammation throughout your body, including any inflammation affecting your myelin.
Tecfidera (dimethyl fumarate) is also an anti-inflammatory substance. It specifically targets inflammation around myelin and protects your nerve cells.
Some DMDs prevent immune cells from crossing the blood-brain barrier, so they cannot get to the CNS to damage your myelin. Tysabri (natalizumab) interrupts the signal that lets immune cells through the blood-brain barrier. Gilenya (fingolimod) traps immune cells in the lymph glands so they can't migrate into the central nervous system.
Other DMDs aim to reduce the numbers of lymphocytes in the body, so that there are less to attack your myelin. Aubagio (teriflunomide) and Mavenclad (cladribine) restrict the numbers of T-cells and B-cells in circulation in your body. Lemtrada (lemtuzumab) kills off all white blood cells that are actively attacking your myelin. Ocrevus (ocrelizumab) targets and destroys B-cells specifically.
Some of the side effects you might find with the different DMDs are a result of their effect on your immune system. For example, if the effectiveness of the immune system is reduced, then dormant diseases might spring up again in your body. The immune system offers a large number of future targets for MS drugs, as we learn more about how the different cells interact.
Stem Cell Transplant or AHSCT aims to reboot your immune system using your own stem cells. Stem cells are able to mature into all different kinds of cells.
In AHSCT stem cells are harvested from your blood, and then your existing immune cells are killed off using aggressive chemotherapy. Your own stem cells are then replaced in your body. The intention is that these fresh stem cells will develop into all the immune cell types required, but be free of the tendency to cause MS.
As the immune cells grow back, you have little protection against infection, which is why care must be taken to keep isolated until your immune system is back to full strength.